

Associate Professor
Department of Biology
Western University
1151 Richmond St. N
London, Ontario, Canada
N6A 5B7
Office: Western Science Center 305
Email: aperciva@uwo.ca
Phone: (519) 661-4015
Fax: 519 661-3935
The development of a multicellular organism from a single celled fertilized egg has long fascinated Biologists. Over the past forty years, the major molecular components that control development have been identified. The interaction between these components has been well characterized, and nowhere better illustrated than the hierarchy of factors that establish the segmented body plan of the fruit fly, Drosophila melanogaster. Many of these factors are transcription factors that regulate the rate of initiation of transcription.
Differential pleiotropy and HOX functional organization
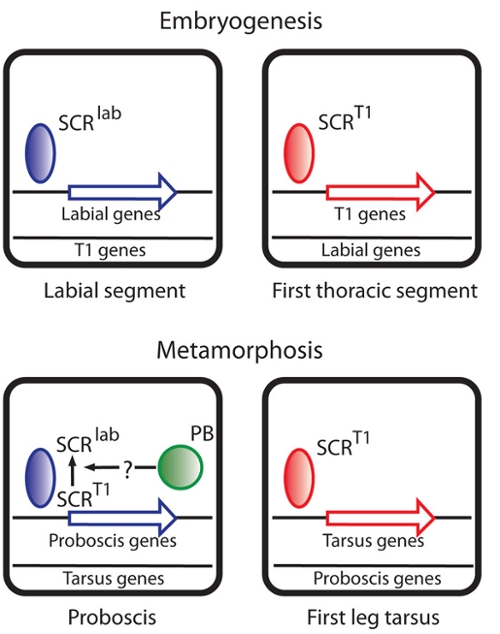
We study HOX transcription factors required for determining the number of segments that form, Fushi tarazu, and factors required for determining the identity/structure that a segment will develop, Proboscipedia and Sex combs reduced. Although most transcription factors are thought to be composed of modular domains that mediate an unique aspect of transcription factor function like DNA binding, transcriptional activation/repression, and binding regulatory factors, the HOX proteins are proving to be quite dissimilar to this view of the archetypic transcription factor in being composed in most cases of small peptide motifs that make small and differential contributions to overall HOX activity. We are interested in how HOX proteins work as transcription factors and what processes they control in developmental programs.
Phenotypic non-specificity
Phenotypic non-specificity is the phenomenon of multiple transcription factors being able to induce or rescue a phenotype. The examples shown below are four transcription factors that recognize distinct DNA-binding sites but are able to induce both wingless and eyeless phenotypes. We are characterizing phenotypic non-specificity further by assessing how common it is in both Drosophila and yeast, and we are trying to demonstrate the phenomenon of cis-element bypass, which is a prediction of the model of limited specificity of transcription factor function that we have proposed to explain phenotypic non-specificity. Phenotypic non-specificity has ramifications for our view of transcription factor function and our view of evolution and development.
